ZB-5a Research Group of Organic Synthesis
Achievements
Research profile
Our research, combining elements of organic synthesis, physical organic and computational chemistry, is focused on the design of new aromatic and heteroaromatic structures of both theoretical and practical interest. We look for new, efficient synthetic methods that will provide access to compounds characterized by non-trivial fusion patterns and often considerable out-of-plane distortions. Our current research interests cover a range of areas of potential relevance to materials chemistry:
- New annulation, heteroannulation and macrocyclization methods, as tools for the synthesis of extensively conjugated aromatics.
- Organic reaction mechanisms, with a focus on coupling reactions.
- Synthesis of π-conjugated heteroaromatic polymers, including heteroatom-doped graphene nanoribbons.
- Active materials for organic photovoltaics.
Highly strained aromatics
Research goals:
- Synthesis of distorted aromatic molecules with exceptionally high strain energies.
- Correlating distortions and strain energies with the electronic structure and reactivity.
- Distorted aromatics as receptors and self-assembling materials.
Key results:
- Development of the fold-in method for the synthesis of chrysaoroles and related molecules (Angew. Chem. 2013, Chem. Eur. J. 2014, Chem. Commun. 2015).
- „Nonclassical nanotube endcaps” – a single-step synthesis of exceptionally strained heterocycles in a synchronized homocoupling (JACS 2015).

Nanographenes and their heteroanalogues
Research goeal:
- Synthesis of extended aromatics with unusual conjugated frameworks.
- Near-infrared chromophores and fluorophores
- Self-assembly in condesed phases.
Key results:
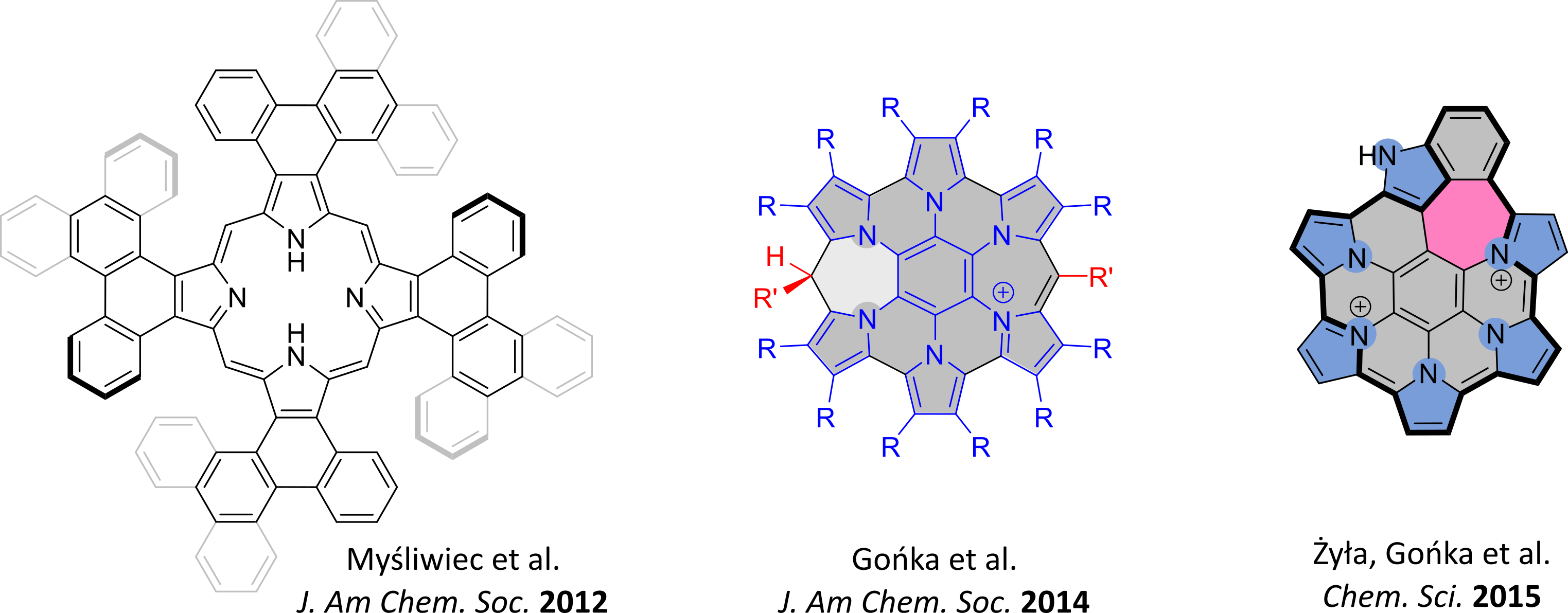
- Heteroaromatic nanographenoids containing 7-membered rings (JACS 2014, Chem. Sci. 2015).
- Liquid-crystalline, peripherally condensed oligopyrroles (JACS 2012).
Synthetic methodology
Research goals:
- Soichiometric and catalytic synthetic methods in macrocyclization reactions.
- Oxidative coupling reactions as a tool for the development of conjugated frameworks.
- Transition metal-mediated reactions as a tool for induction of internal strain.
Key results:
- The fold-in strategy, synchronized homocpoupling, aromatic oxidative coupling (examples above).
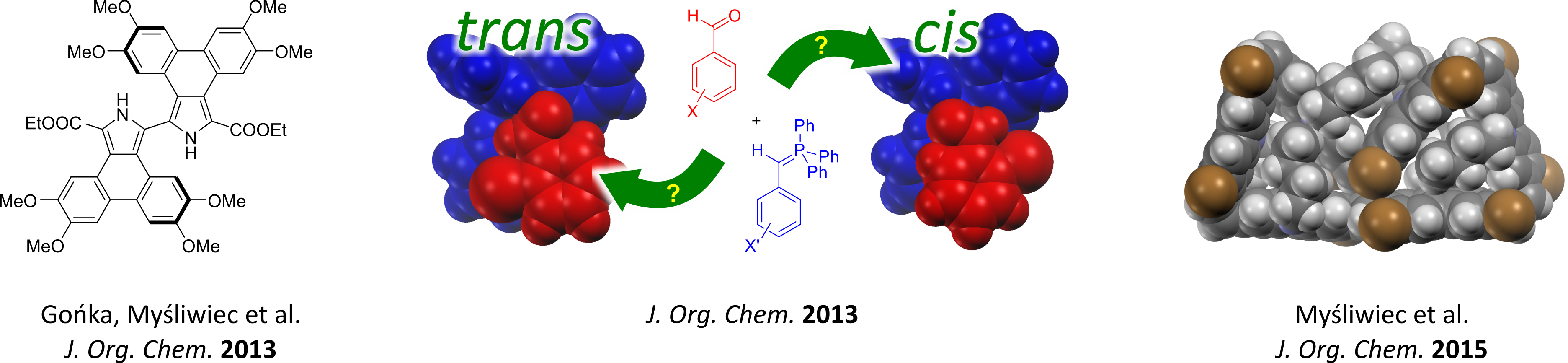
- Tandem oxidative coupling for the synthesis of phehanthropyrroles (JOC 2013).
- Theroretical investigations of the Wittig reaction (JOC 2013).
- Synthesis of expanded carbazolophanes in a stereoselective Wittig reaction (JOC 2015).
 Nanographenes and their heteroanalogues
Research goeal:
Nanographenes and their heteroanalogues
Research goeal:
 Synthetic methodology
Research goals:
Synthetic methodology
Research goals:


